Societal Implications and Applications
There are many societal implications and applications that can be related to this concept Listed below are some that can be addressed given in terms of relation to lesson subtopic along with a link to possible teaching ideas for incorporating this aspect of the curriculum.
Smog Warnings and Brown Skies-Le Chatelier's Principle
|
The reaction that occurs on how sunny days that leads to brown skies and smog warnings can be explained quite easily using the knowledge of Le Chatelier's principle. The reactions that occur are listed below:
1. N2 + O2 –> 2NO Nitric oxide can then combine further to produce nitrogen dioxide 2. 2NO + O2 –> 2NO2 Nitrogen dioxide can then dimerize to form dinitrogen tetroxide. 3. 2NO2 <–> N2O4 + heat This is an exothermic reaction, that is to say that in this direction heat is released. Dinitrogen tetroxide is a colourless gas-it is the nitrogen dioxide that turns our sky brown! So on a hot sunny day, when the temperature rises the equilibrium between NO2 and N2O4 shifts in the direction of NO2 to combat the increase of temperature and the sky turns brown! |
|
The Haber Process-Le Chatelier's Principle
|
The Haber process is an industrial process used to produce ammonia that makes use of the properties of Le Chatelier's principle to maximize yield. Its importance in society is paramount-the fertilizer made from ammonia is used to sustain over one third of the world's population. There are numerous reasons for the controversy surrounding this industrial process. One of the most notable is the creation of NH4NO3 which relies on the use of fossil fuels, thus our food supply is highly dependent on resources that may be limited. Another good choice for student research would be into the impact that the creation of mass amounts of NH4NO3 have on the degradation of water and soil resources and possible solution or alternative synthesis pathways that may benefit society in the long run. |
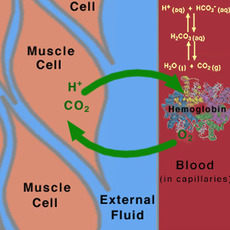
Breathing-Buffers
|
This is an application of buffers at its finest. Carbon dioxide is toxic in the blood if kept in its natural form. So how does the body naturally get rid of this toxic gas? The body has its own buffering system to allow carbon dioxide to flow through the body without any serious and drastic side effects. The equation for the bodies bicarbonate system is given below: CO2 + H2O ↔ H2CO3 ↔ HCO3- + H+ As soon as the carbon dioxide is released into the blood, it mixes with water to create carbonic acid which is then converted to its conjugate bicarbonate ion. This buffering systems prevents huge fluctuations in pH that would be potentially fatal. |
Stalactites and Stalagmites
Please click below for an example of a culminating assignment that can be used to introduce many of the societal implications and applications of this strand of the curriculum.